



Initiated by VEMINSYN, the group standard T/SHRH 064-2024 Transdermal Permeation Test of Cosmetic Ingredients – In Vivo Raman Spectroscopy on Human Body was jointly developed by 26 organizations (listed in no particular order), including:Proya, Shanghai Jahwa, NIVEA, Unilever, HORIBA, Microtest, SGS, Coty, LAN, Kans, Yuanxiang Bio, Huanya Group, Clorisen, Turtle Dad, PMPM, Nice, Rongda Bio, Youwei Bio, Nosibell, Femeller, Bloomage Biotech, Mistine, Shenzhen Academy of Metrology and Quality Inspection, Shanghai Chuangyuan, and Xiaochuang Testing.

Currently, no industry standard has been established for testing the penetration behavior of cosmetic active ingredients on in vivo human skin, which has restricted the wider application of cosmetic active ingredients to a certain extent.
This standard is the first group standard in China for in vivo human skin testing and evaluation methods of active ingredients in the cosmetics field.It defines the basic principles, test mechanism, test procedure, result calculation and test report content for the transdermal permeation evaluation of cosmetic ingredients using in vivo Raman spectroscopy on human skin.

*Above:LabRam Soleil
Standards guide excellence, quality defines achievement.To foster the sound development of the cosmetic active ingredients industry, the in vivo human skin Raman testing method proposed by VEMINSYN has filled this industry gap. As the pioneer leading the research of this group standard, VEMINSYN joined hands with numerous leading enterprises and institutions across the sector to complete its development. This standard stands as one of the most influential group standards in the industry in terms of participating organizations.
Meanwhile, the release of Transdermal Permeation Test of Cosmetic Ingredients – In Vivo Raman Spectroscopy on Human Body further clarifies the testing protocols, basic principles, systematic procedures and detailed specifications for applying Raman spectroscopy in in vivo human tests. It provides technical guidance for evaluating the transdermal penetration behavior of cosmetic active ingredients via Raman spectroscopy.
The launch of this standard not only fills the domestic vacancy for in vivo human Raman spectroscopy testing in cosmetics, but also lays a solid foundation for the advancement of novel active ingredients in the cosmetic industry.
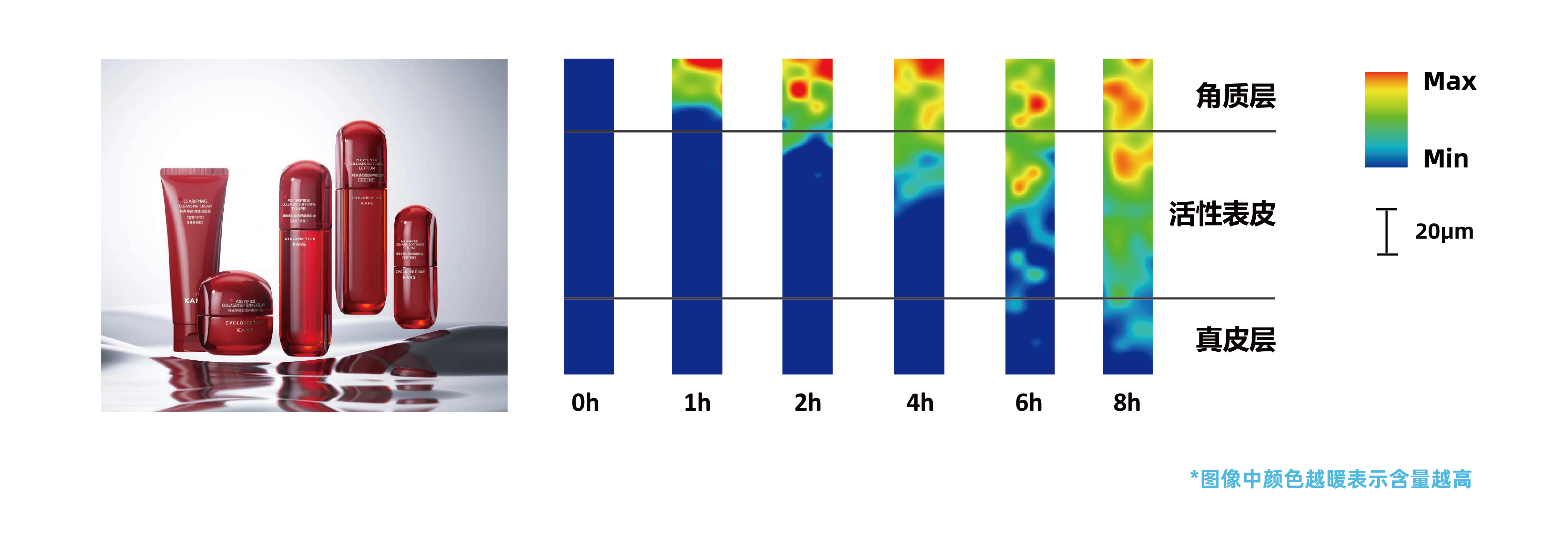
*Above: In vivo human Raman test results of Kans Peptide Collagen Lifting & Firming Essence
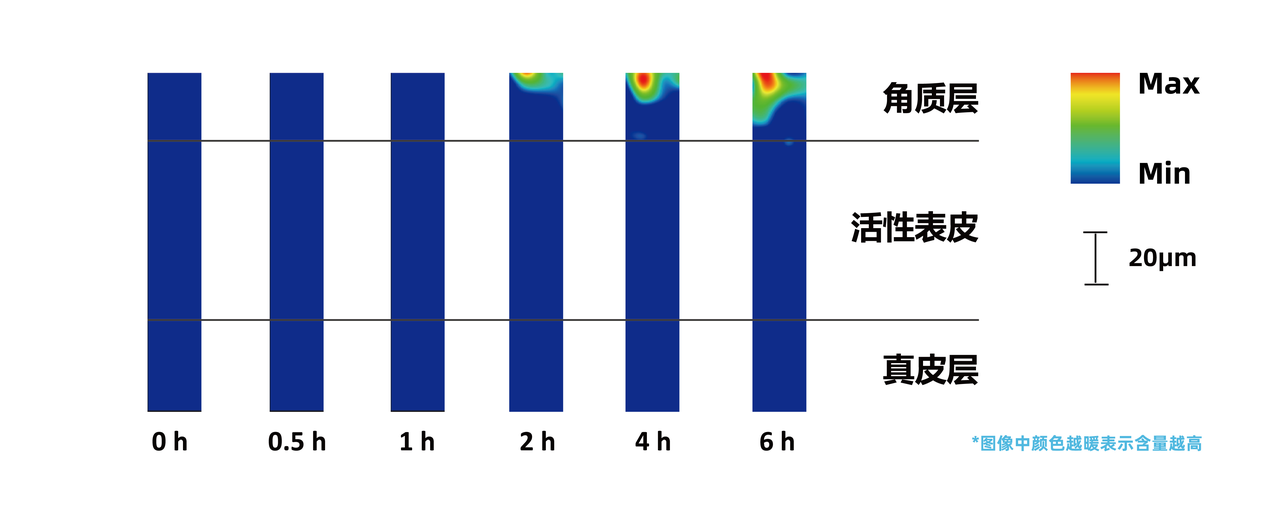
*Above:In vivo human Raman test results of sunscreen agents in a commercially available sunscreen gel



